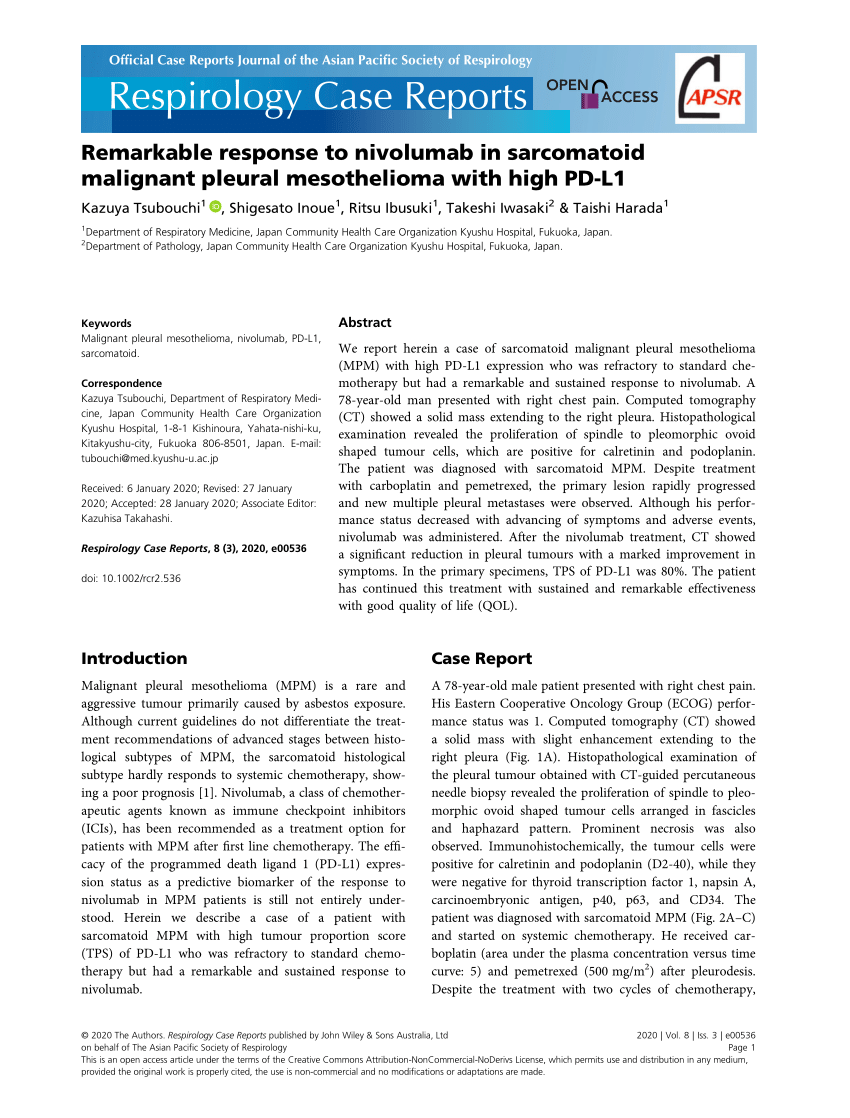
Nivolumab And Mesothelioma, Fda Approves Bms Opdivo And Yervoy Combination In First Line Mesothelioma
Nivolumab and mesothelioma Indeed lately is being sought by consumers around us, perhaps one of you personally. Individuals now are accustomed to using the net in gadgets to view image and video information for inspiration, and according to the name of this post I will talk about about Nivolumab And Mesothelioma.
- Https Oncologie Mumc Nl Sites Oncologie Files Sclc Mesotheliomen Pdf
- Confirm Mesothelioma Research Programme Leicester
- Onclive Com On Twitter Paul Baas Md Phd Of Nki Nl Discusses The Combination Of Nivolumab Plus Ipilimumab Significantly Improved Overall Survival Versus Chemotherapy In The First Line Treatment Of Patients With Unresectable Malignant Pleural
- Chemo Free Regimen Boosts Os In Mesothelioma Medpage Today
- Ipilimumab And Nivolumab In The Treatment Of Recurrent Malignant Pleural Mesothelioma Initiate Results Of A Prospective Single Arm Phase 2 Trial The Lancet Respiratory Medicine
- Pdf Confirm A Double Blind Placebo Controlled Phase Iii Clinical Trial Investigating The Effect Of Nivolumab In Patients With Relapsed Mesothelioma Study Protocol For A Randomised Controlled Trial
Find, Read, And Discover Nivolumab And Mesothelioma, Such Us:
- Pairing Immunotherapy And Virotherapy For Mesothelioma Mesothelioma Guide
- Bristol Myers Squibb S Opdivo Yervoy Combo Helps Mesothelioma Patients Live Longer Fiercepharma
- Nivolumab Plus Ipilimumab Improves Survival In Frontline Mesothelioma Onclive
- Fda Approves Nivolumab And Ipilimumab For Malignant Pleural Mesothelioma Ons Voice
- Nivolumab And Ipillimumab Show Promise In Mesothelioma Mesothelioma Com
- Pinkalicious And Peterrific Pinkalicious Coloring Pages
- What Does Asbestos Tile Adhesive Look Like
- Ram Coloring Page
- Uber Accident Lawyers
- Naming A Law Firm
If you re searching for Naming A Law Firm you've arrived at the ideal place. We ve got 100 images about naming a law firm adding pictures, photos, photographs, backgrounds, and more. In such web page, we also have variety of images available. Such as png, jpg, animated gifs, pic art, logo, black and white, translucent, etc.
Research suggests that 3 out of 4 mesothelioma tumors overexpress pd l1.
Naming a law firm. Nivolumab and ipilimumab represent a new treatment possibility shaping the options available to treat difficult diseases like mesothelioma. Today the fda approved opdivo nivolumab in combination with yervoy ipilimumab for the first line treatment of adults with malignant pleural mesothelioma that cannot be removed by surgery. Researchers in germany are recruiting patients for a trial of nivolumab and chemotherapy for pleural mesothelioma patients after surgery.
Researchers hope that nivolumab or the combination of nivolumab with ipilimumab may someday become a proven second line or third line treatment method for pleural mesothelioma. By next fall researchers anticipate to collect more data from the maps 2 trial including more information about mature survival quality of life and biomarker data. The goal of the new study was to provide real world data on the treatment.
The merit study was a single arm phase ii clinical trial of nivolumab for the second or third line treatment of patients with malignant pleural mesothelioma in japan. Dual checkpoint inhibition with nivolumab and ipilimumab is associated with prolonged overall survival os relative to chemotherapy in treatment naive patients with inoperable malignant pleural mesothelioma show checkmate 743 data. Randomized phase iii of nivolumab for mesothelioma are ongoing.
Standard mesothelioma treatments include surgery chemotherapy and. This is the first positive randomized trial of dual immunotherapy in the first line treatment of patients with mesothelioma said the. The study included 125 patients 80 percent of whom were male with a median age of 72 years old.
The recommended doses for unresectable malignant pleural mesothelioma are nivolumab 360 mg every 3 weeks and ipilimumab 1 mgkg every 6 weeks until disease progression unacceptable toxicity or. All participants had advanced malignant pleural mesothelioma had received zero to two mesothelioma treatments before and were randomly assigned to receive either just nivolumab or a combination of nivolumab with ipilimumab. When they convince the immune system to work against the cancer the positive effects can last much longer than traditional chemotherapies.
It is the same protein blocked by the immunotherapy drug keytruda.
More From Naming A Law Firm
- Entertainment Law Firms Miami
- Halloween Face Painting Images
- Mesothelioma And Cms Collections
- Dechert Offices
- Mesothelioma Testicular Pain
Incoming Search Terms:
- Anti Angiogenic And Immunotherapeutic Approaches In Mesothelioma Memoinoncology Mesothelioma Testicular Pain,
- Pdf Nivolumab Induced Immune Thrombocytopenia In A Patient With Malignant Pleural Mesothelioma Mesothelioma Testicular Pain,
- A Phase Ii Trial Of First Line Combination Chemotherapy With Cisplatin Pemetrexed And Nivolumab For Unresectable Malignant Pleural Mesothelioma A Study Protocol Sciencedirect Mesothelioma Testicular Pain,
- Nivolumab Plus Ipilimumab Improves Os In Malignant Pleural Mesothelioma Cancer Network Mesothelioma Testicular Pain,
- Bristol Myers Squibb Releases Optimistic Interim Results In Mesothelioma Clinical Trial Mesothelioma Applied Research Foundation Mesothelioma Testicular Pain,
- Onclive Com On Twitter Paul Baas Md Phd Of Nki Nl Discusses The Combination Of Nivolumab Plus Ipilimumab Significantly Improved Overall Survival Versus Chemotherapy In The First Line Treatment Of Patients With Unresectable Malignant Pleural Mesothelioma Testicular Pain,








